At FyoniBio, we know that every biotherapeutic journey is unique, and so are the solutions it requires.
Our integrated upstream (USP) and downstream (DSP) process development services are designed to help you advance your recombinant protein from concept to clinic with confidence.
We identify the right cell line for your molecule, boost productivity, and design a high-yield purification process—guided by deep analytical expertise and cross-functional insight.
With a strong track record across mAbs, bispecifics, fragments, fusion proteins, enzymes, and other complex or glyco-optimized biologics, we help you achieve scalable expression, robust reproducibility, and smooth tech transfer to GMP partners in the US and Europe.
Why choose us
End-to-end USP and DSP ready for tech transfer and scale-up to pilot or production level
Optimization of existing processes to boost efficiency and product quality
Control strategy implementation aligned with GMP/ICH guidelines
Application of Design of Experiment (DoE) methodologies and robustness testing
Formulation development and stability studies under R&D conditions
Development of customized analytical assays and bioassays
Protein modification and conjugation services
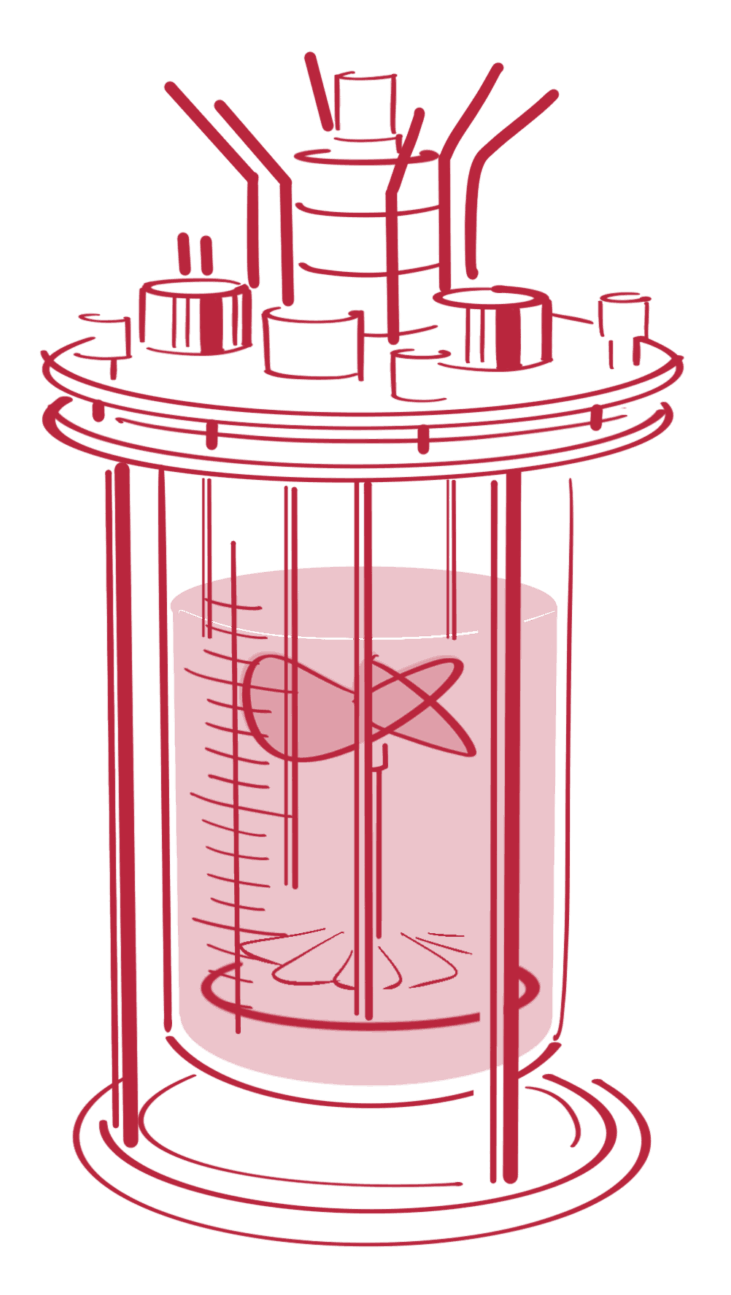
Our upstream process development leverages robust mammalian cell culture systems, including our proprietary GlycoExpress® and CHOnamite® platforms. We combine flexible process modes—batch, fed-batch, intensified fed-batch, and perfusion—with expert-driven optimization to help your biotherapeutic advance from early development to GMP manufacturing.
Capabilities Include:
PROCESS
01
A simple, one-step process. Nutrients are added at the start, and the culture runs its course without further intervention—ideal for early-stage studies or robust cell lines.
02
A controlled way to boost yield. Nutrients are added gradually, allowing for longer cultivation and higher productivity compared to batch mode.
03
An optimized version of fed-batch. By fine-tuning feeding strategies and process parameters, it supports higher cell densities and more efficient production.
04
A continuous process that keeps cells in a steady state. By constantly refreshing the medium, perfusion enables very high productivity and consistent product quality—especially useful for challenging proteins.
PROCESS
Batch
A simple, one-step process. Nutrients are added at the start, and the culture runs its course without further intervention—ideal for early-stage studies or robust cell lines.
Fed-batch
A controlled way to boost yield. Nutrients are added gradually, allowing for longer cultivation and higher productivity compared to batch mode.
Intensified Fed-batch
An optimized version of fed-batch. By fine-tuning feeding strategies and process parameters, it supports higher cell densities and more efficient production.
Perfusion
A continuous process that keeps cells in a steady state. By constantly refreshing the medium, perfusion enables very high productivity and consistent product quality—especially useful for challenging proteins.
Our downstream process development focuses on preserving the structural and functional integrity of your protein while effectively eliminating critical process-related impurities such as host cell proteins (HCP), DNA, and media components.
Capabilities Include:
Throughout all stages of process development, our integrated analytical services & bioassays department provides the critical data you need to move forward with confidence. Whether in upstream optimization or downstream purification, our analytics ensure quality determination, in-depth characterization, and regulatory readiness for your therapeutics.
Backed by decades of experience and ISO 9001-certified systems, we offer fully customized or newly developed assays to meet the unique requirements of your project. Our analytical capabilities include:
We also offer:
As part of our comprehensive process development services, we offer formulation development and targeted protein modification. Protein stability can be compromised during long-term storage due to aggregation, fragmentation, deamidation, or oxidation. To mitigate these risks, we apply accelerated stress studies and buffer screening, using intrinsic or dye fluorescence monitoring, to optimize storage conditions for both liquid and lyophilized formulations.
To further support your project, we provide a range of protein modification solutions—offering conjugation services such as ADCs, streptavidin, biotin, fluorescent labels, and more. We also develop custom affinity matrices by coupling your protein to NHS-activated resin or magnetic beads.
Case Study
Developing a Cost-Effective Purification Process for a Hydrophobic Bispecific IgG2 Antibod.
At FyoniBio, upstream and downstream process development can be performed and adapted to provide a customized solution for each project. From upstream development several small-scale models are available to mimic large scale production processes. From simple shake flask and SPINtube models up to well monitored AMBR15 small scale bioreactors, optimization strategies for fed-batch and perfusion processes can be performed.
The FyoniBio staff has many years of experience in process development and optimization for antibodies, bispecific antibodies, blood factors and other glycoproteins of their internal pipeline as well as of customer projects. Starting from affinity capture, one or two polishing steps using ionic, hydrophobic or mixed mode chromatography lead to very pure products with lowest HCP, HCD, and aggregate content. In addition, formulation development and stability studies find buffers suitable for long-term storage of the protein.
During a perfusion process, the product is constantly removed from the bioreactor while fresh media is fed. In contrast, in a fed-batch process the product stays over 14-17 days in a bioreactor at cell cultivation conditions (37 °C, high shear forces). If products are unstable over fed-batch cultivation time, perfusion processes might be the process of choice as harvested supernatant can be directly stored at the required storage conditions. Apart from the fact of instability, posttranslational modifications can be kept constant over the time period the perfusion process is run by continuously holding cells in a constant state.
Protein purification processes consist of several steps. Optimally, an affinity chromatography resin is available, such as Protein A for immunoglobulins, since it results in very pure target protein. One or two polishing steps follow, which can be ion exchangers, hydrophobic interaction or mixed mode resins. Often the buffers have to be exchanged or adapted between steps.
The goal of each downstream process is to get rid of host-cell proteins and DNA, eliminate possible virus contamination, and deplete aggregated or inactive target protein. Preserving functionality must always be kept in mind.
For classical IgG1 molecules a platform downstream process is available which is adjusted to the individual mAb in a short and cost effective manner.
A protein is a very complex molecule. Therefore, several methods have to be used to fully characterize it. Methods analyzing amino acid composition, glycosylation or other post-translational modifications are described in the section about mass spectrometry services. However, intact mass, aggregates, fragments, net charge, identity and isoforms can also be detected by other methods:
The biochemical and biophysical protein characterization is complimented by protein- and target specific functionality testing (e.g., target binding, signaling, cell-based assays). Learn more about functional bioassays.
At FyoniBio, we have long-term experiences in the characterization of proteins with a broad spectrum of analytical methods. Contact us to discuss the specific needs of your project and how we can support you.
To enable a fast and accurate process transfer a technology transfer report is written including all information about the developed processes. Technology transfer reports can be directly delivered to a selected CMO. FyoniBio offers additional services to support the process transfer by implementation of process transfer meetings and direct on-site process support.
Successful tech transfer processes have been performed to different GMP sites around the world.
Our experienced team is here to provide the analytical support and insights
you need to move your program forward efficiently, confidently, and without delays.